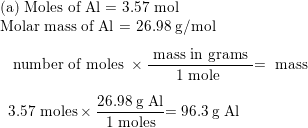The Mole. The Mole The Mole Memorize this number: 1 mol = 6.02 x 1023 of something A mole is defined as the number of particles in exactly 12g of Carbon ppt download

The Mole. What is the a mole? A unit to measure the amount of a substance. Commonly abbreviated mol. The symbol is n. - ppt download

SOLVED:Perform the following conversions a. 1.51 X 1015 atoms of Si to mol of Si b. 4.25 X 10-2 mol of H2SO4 to molecules of HzSO4 c: 8.95 X 1025 molecules of